Vitamin D Hair Loss Reversal
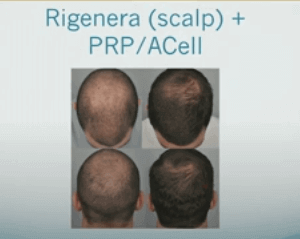
We have heard about the Regenera Activa® (also spelled Rigenera) hair loss treatment regularly since 2019.
I briefly mentioned it in several posts, including in Dr. Cooley's presentation that I discussed here. Many doctors use both Regenera and platelet-rich plasma (PRP) together. Both procedures are autologous in nature and therefore unlikely to cause any major side effects in the hands of experienced practitioners.
At least 20 people have commented about this stem cell based treatment over the years. It is far more popular in Europe and Asia than in the US, although the Regenera device is approved for use by the US FDA.
I was not keen to write an entire post on the product as yet. For one, online reviews and forum testimonials are all over the map, and lean towards significant skepticism. However, things are getting more interesting now.
Regenera Activa AMT
Most new hair loss technologies and companies tend to see diminished social media presence over the years. Most also stop updating their websites or make sporadic changes. For a while, I thought Regenera Activa was becoming stagnant. However:
-
- Since my last visit over a year ago, Regenera has made a complete overhaul of its website. It seems like the Rigenera spelling is now being avoided.
- They now offer a pdf that recommends using the Regenera Activa treatment alongside a hair transplant procedure.
- They have re-branded their product as AMT® . An acronym that stands for Autologous Micrografting Technology.
- A new November 2021 study from Saudi Arabia concludes that Regenera Activa benefits hair growth positively. In both males and females; and in both hair quantity and hair thickness. Note that this technology is described as Autologous Cellular Micrografts (ACM).
- Also in 2021, Dr. Antonella Tosti (Italy) made a video webinar presentation on AMT Regenera. It includes before and after positive hair growth results from a small study of 17 people. Her patients on average saw a significant increase in frontal hair thickness, but no significant increase in hair count.
- The Spain based company maintains regularly updated Instagram and Facebook pages.
- The number of hair transplant surgeons offering Regenera Activa stem cell treatment is growing very rapidly. The latest video from yesterday comes all the way from Pakistan:
How Does it Work?
As seen in the above video, the Regenera Activa method involves three main steps.
- The first stage entails extraction of of 3-5 small scalp skin samples using a 2 mm punch biopsy. These extractions are collected from the occipital region of the scalp that is least susceptible to androgenic alopecia. i.e., from the permanent donor zone area that is also utilized during hair transplant procedures.
- The extracted skin samples are then placed in the Rigeneracon device. They undergo centrifugation at a speed of 80 rpm for 2-6 minutes. The resulting filtered solution contains stem cells, growth factors, extracellular matrix and progenitor cells.
- Finally, the resulting mixture is injected into the scalp. The mixture of stem cells and growth factors is though to help shrinking hair follicles in balding areas to regenerate.
Pricing
The price of one session of Regenera Activa hair loss treatment varies by country and clinic. However, the range is not as wide as is the case with PRP. Note that a surgeon might offer it at a steep discount if you are also getting a hair transplant with him or her. According to the Daily Mail, the UK price at one clinic is £2,000. In the US, most clinics seem to be advertising the price at $3,000.
I first bookmarked a page about this procedure from Dr. Ee in Singapore in 2018. You can see him in this video.
Kintor Pharmaceutical (China) is definitely the real deal. They are moving faster than any other company in the history of the hair loss industry. I think all my readers will agree.
Kintor will Start Phase III Trials in China in January 2022
In the latest update from today (November 24th, 2021), Kintor Pharmaceutical announced that it will begin Phase 3 trials in China in January 2022!
The company's IND application for the pivotal study (phase III) of pyrilutamide (KX-826) was cleared by China's National Medical Products Administration. KX-826 is the first androgen receptor (AR) antagonist to enter phase III clinical trials anywhere in the world.
This is perhaps the best news in the hair loss world so far in 2021.
Update: November 12, 2021 — Kintor Pharma just started Phase II clinical trials in women in China with a first dose of pyrilutamide (KX-826). This trial pertains to androgenetic alopecia in women (i.e., female pattern hair loss).
Update: September 8, 2021 — Kintor Pharma just announced that its Phase II clinical study for KX-826 to treat androgenetic alopecia was a success. Primary endpoint was met. More detailed results will be released later per CEO Dr. Youzhi Tong. Phase III clinical trials in China will commence in the fourth quarter of 2021. Even better, Phase II trials are also currently taking place in the US. And Phase II trials for women will take place in China
Update: July 11, 2021 — The US FDA just approved Kintor's Phase II clinical trial for pyrilutamide (KX-826) to treat androgenetic alopecia. Note that the company's trials in China are already mid-way through Phase II per their pipeline page.
They area also working on another hair loss product named GT200029 that is an "AR-PROTAC" compound. Its Phase 1 trials will start in China this month.
April 15, 2021
Kintor Pharma: AR Antagonist and AR Degrader
Earlier today, it was announced that Kintor received approval in China to begin clinical trials for GT20029. This product will be in tincture or gel format, and will be tested for the treatment of androgenetic alopecia and acne.
- The GT20029 product is an androgen receptor degrader (AR Degrader). It is developed using Kintor's proprietary Proteolysis Targeting Chimera (PROTAC) platform. According to the press release, this is the world's first topical androgen receptor (AR) compound (AR-PROTAC) to enter clinical trials. GT20029 degrades the AR protein via the E3 ubiquitin ligase pathway. During preclinical studies, GT20029 did not cause any notable side effects or systemic drug accumulation.
- Note that Kintor's main product for treating male pattern hair loss is KX-826 (Pyrilutamide) and is an androgen receptor antagonist (AR Antagonist). I covered the latter in prior updates to this post if you read till the end. KX-826 is currently in Phase 2 clinical trials in China and in Phase 1 trials in the US.
Note that Cassiopea's Breezula (Clascoterone) is an AR antagonist that is well ahead of KX-826 when it comes to clinical trial stage. Kintor's website has a very interesting article discussing both AR antagonist products and hair loss in China in general.
Make sure to also read my related past post on destroying the androgen receptor to reverse hair loss.
Feb 2, 2021
Clinical Trial Status
Kintor's investigational new drug (IND) application of GT20029 for androgenetic alopecia and acne vulgaris was accepted by the National Medical Products Administration (NMPA) of China.
Kintor has moved forward with its trials faster than any other hair loss company. I am glad to see a Chinese company finally entering the hair loss cure market. Scientific and technological progress seem to happen faster in China than in the west. Hopefully, clinical trials for hair loss products will follow the same pattern.
Side note: In July 2020, Kintor and Applied Biology (US) collaborated on using Proxalutamide for the Treatment of COVID-19. There is a school of thought that suggests anti-androgens could help reduce Coronavirus fatalities. To date, more men have died from the disease then have women.
Below is the pipeline from Kintor's website:
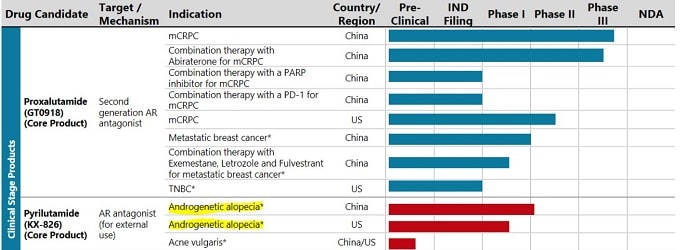
Kintor Pharmaceutical (China) also recently completed the enrollment of 120 patients in its Phase II clinical trials for pyrilutamide for hair loss. See the bottom half of this post for my original discussion on Kintor. Their stock is traded on the Hong Kong Hang Seng Index.
Key quote from CEO Dr. Youzhi Tong:
"We will accelerate the progress of its phase II/III clinical study so as to bring benefits to the people suffering from alopecia as soon as possible."
May 26, 2020
A new Chinese company named Kintor Pharmaceutical is working on an interesting hair loss drug called pyrilutamide . It is extremely rare to hear about any Chinese company involved in hair loss cure research. Very strange, considering the country's rapid pace of scientific advancement and massive population. Moreover, Chinese men and women are nowadays balding at much faster rates than in the past.
Update: August 4, 2020 — Phase Ib trials are now complete.
Kintor Pharmaceutical and Hair Loss
Four days ago, China-based Kintor Pharmaceutical (also known as Suzhou Kintor Pharmaceuticals) got significant Chinese media coverage. This interest was related to the company's prostate cancer, breast cancer and hair loss drugs.
Earlier this month, Kintor Pharmaceutical also had a very successful IPO in Hong Kong.
While the company's main focus seems to be its prostate cancer and breast cancer drugs, its androgenetic alopecia drug trials are also advancing rapidly. Their main androgen receptor blocking drug candidate is called Pyrilutamide (KX-826) and it is applied to the scalp topically. The company's Proxalutamide drug slows or stops cancer cell growth by entirely inhibiting androgens.
Pyrilutamide
The one disappointing news is that Kintor aims to take on Johnson & Johnson's Minoxidil. This could mean that topical Pyrilutamide is unlikely to be much better than Minoxidil. I hope I am wrong. Recently completed phase one trials in China proved that pyrilutamide is safe and causes no major side effects in humans.
Kintor is currently conducting phase 2 clinical trials for pyrilutamide on 160 men in China, and phase 1 trials on 30 men in the US. Phase 3 trials on 600 people in China, the US and Japan are planned for as soon as 2021. I would guess that the US FDA and Japanese PMDA will never accept Phase 2 results from China as any kind of proof to proceed to Phase 3 trials in the US and Japan.
So how can the company proceed so fast in the US and Japan?
Source: https://www.hairlosscure2020.com/







Tidak ada komentar:
Posting Komentar